 Here is a complete list of alkali metals present on the Periodic table with their atomic number, symbol and name of element. Cesium has high reactivity, but it is predicted that Francium would have even higher reactivity than that of cesium)Īlso visit: Different types of metals on periodic table. And hence for any practical purposes, cesium is considered as the most reactive metal in Periodic table. ( Note: Francium is a laboratory made element. Thus as the size of an atom increases, the loss of electron becomes very easy.Īnd hence the element with bigger atomic size (i.e Francium) is most reactive alkali metal. Also alkali metals have only one electron in their outermost orbit. Look, as we move down the group from top to bottom, the atomic size increases. Due to less intermolecular force and weak metallic bonds, they have more tendency to react with other elements. They have less intermolecular attractive force and have less density. #4 Lower boiling and melting point | Less density | Weak metallic bondsĪlthough alkali metals are solids, they have lower boiling point and melting point. 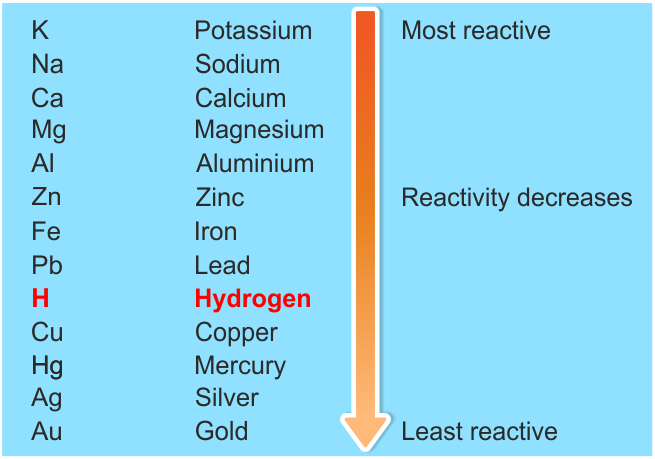
So chemical reactions take place very fast and thus they are more reactive.Īlso read: Ionization energy trend in periodic table. Thus less ionization energy indicates that the atoms show a high tendency to lose electrons. And as we move from left to right, the ionization energy increases. Now this ionization energy is very less for the elements located on the left side of the Periodic table. Ionization energy is the minimum energy required to remove an electron from the gaseous atom or ion. #2 One valence electronĪlkali metals are present in the first group of Periodic table.įirst group indicates that they have only one electron in their outermost orbit.Īs only one electron is present in the outermost orbit, it is very easy to lose or donate this electron during a chemical reaction.Īs it is easy to donate the electron, the chemical reaction is very easy and this makes alkali metals more reactive.Īlso read: Valency trend in periodic table. This makes them highly reactive.Īlso read: Atomic size trend in periodic table. Larger atomic size of alkali metals indicates that there is a less attractive force between the nucleus and outermost electron.ĭue to less attraction force, the alkali metals show a tendency to lose electrons easily. 
There are many reasons behind the reactivity of alkali metals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
